VNS COVID-19 White Paper
Transcutaneous Auricular Vagus Nerve Stimulation for the Management of COVID-19 Infection
Background
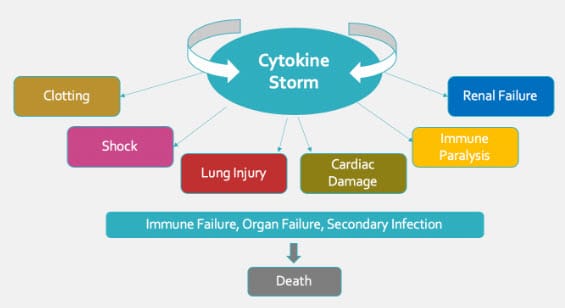
Patients hospitalized with COVID-19 predominantly die of organ failure due to a surge of pro-inflammatory cytokines triggering the need for mechanical ventilation, often due to acute respiratory distress syndrome (ARDS) 1.
Known as a cytokine storm, the surge in cytokines is similar to the excessive inflammatory reaction associated with septic shock (see Figure 1) 2-5.
Anti-viral agents will most likely be required to reduce the molecular viral burden in COVID-19 patients, but an additional approach to control the damaging cytokine release is required to alter the course of disease in hospitalized patients and improve chances of survival (see Figure 2).
Immunosuppressant drugs may reduce inflammation and the tissue damaging cytokines, but they could also be detrimental by inhibiting natural anti-viral immune responses (i.e., suppression of interferons), thereby delaying viral clearance and increasing the risk of secondary infections and death 6-8.
There are two case reports of individuals recovering from COVID-19 pneumonia despite continuing immunosuppressive therapies for renal transplantation (tacrolimus, mycophenolic acid, and prednisone) and breast cancer (fulvestrant and abemaciclib) 9,10. The breast cancer patient’s husband was simultaneously hospitalized with COVID-19 pneumonia and described as being in good health, yet he experienced a worse clinical course (he required admission to ICU, she did not) and longer hospitalization (24 d vs 6 d) than his wife who was simultaneously treated for cancer 10.
The reason for this major difference in disease course may lie in the regulation of the immune system by the vagus nerve. The vagus nerve is involved in an inflammation controlling reflex similar to the blood pressure regulating baroreflex. The vagus inflammatory reflex is triggered when the afferent vagus nerve senses inflammatory products through peripheral receptors (see Figure 3). Vagus nerve activity is relayed through the central nervous system to the efferent vagus nerve. This pathway involves the splenic nerve, which when activated releases norepinephrine and results in suppression of proinflammatory cytokine production by macrophages and alleviates inflammation in many pathological settings (e.g., endotoxemia, peritonitis, or acute kidney injury) 11.
Electrical stimulation of the vagus nerve using VNS can improve the body’s natural ability to regulate the inflammatory response and may be potent enough to suppress pro-inflammatory cytokines and prevent death from COVID-19, especially if used early enough in the course of hospitalization 12-17.
In rat models of sepsis, VNS attenuates the release of pro-inflammatory cytokines, prevents hypotension, modulates coagulation, and prevents fibrinolysis activation, decreasing organ dysfunction, and improving survival 18. Human studies also demonstrate that VNS suppresses the production of proinflammatory cytokines and improves clinical symptoms in rheumatoid arthritis, intractable epilepsy, atrial fibrillation, and Crohn’s Disease 13,16,19,20. This suggests that VNS may be effective in treating disorders characterized by cytokine dysregulation and that it has the potential to prevent hospitalized patients with COVID-19 from progressing to respiratory failure and death.
In my clinical experience of using taVNS over the last 5 years, I have witnessed how taVNS positively impacts inflammation modulation in patients with autoimmune disorders such as rheumatoid arthritis, ankylosing spondylosis and inflammatory bowel disease.
VNS Methodology
The vagus nerve can be electrically stimulated with an implantable device placed on the vagus nerve or by stimulating the auricular branch of the vagus nerve with a conductive surface in contact with the concha of the ear (transcutaneous auricular VNS; taVNS). The taVNS approach is painless, provides easy access to the vagus nerve, and is easy to operate. taVNS has been successfully and safely utilized in over 1000 patients within Dr. Nemechek’s private medical practice.
A transcutaneous approach over the skin of the neck (tnVNS) is also possible but requires an electrical impulse strong enough to penetrate the skin and muscle over the vagus nerve on the lateral aspect of the neck. This makes the stimulation occasionally painful and it is difficult to precisely locate the vagus nerve with this method, making taVNS the preferred method for transcutaneous VNS.
All three approaches of VNS are safe in humans. The transcutaneous auricular approach is the least expensive, does not require surgery, is painless, and makes it easy to precisely target the vagus nerve. It also requires little training and a single unit can be shared among many patients if properly disinfected.
taVNS Provides Needed Immune Regulation
There are no proven therapies against COVID-19 infection other than supportive treatment of the comorbid complications (ARDS, heart failure, renal failure, secondary infections, etc.). A more successful therapeutic approach will most likely involve a combination of supportive care, anti-viral therapy, and suppression of the excessive cytokine release observed in some patients with moderate to severe COVID-19.
Recent treatment guidelines issued by the NIH review the addition of host modifiers/ immunotherapy in moderate to severe COVID-19 patients 21. None of the therapies presently available demonstrate enough efficacy or evidence to garner even a weak recommendation while others, such as JAK inhibitors, are avoided due to their potential to severely compromise the immune system.
A safe option for improved inflammation modulation is transcutaneous auricular vagus nerve stimulation (taVNS) (Figure 5).
In human and animal studies, VNS suppresses pro-inflammatory cytokines and alters the clinical course in severe inflammatory conditions 13,16,19,20. This suggests that VNS may be effective in altering the clinical outcome of hospitalized COVID-19 patients. taVNS is safe, easy to operate, inexpensive, and could be deployed rapidly in large numbers if found to be successful.
Effectiveness of transcutaneous auricular VNS in inflammation modulation
Both implantable and transcutaneous VNS has been extensively studied in animals, with more recent studies in humans confirming its safety and effectiveness. In rat models of sepsis, VNS attenuates the release of pro-inflammatory cytokines, prevents hypotension, and modulates coagulation and fibrinolysis, thus improving survival 23-25. This suggests that VNS has the therapeutic potential prevent hospitalized patients with COVID-19 from progressing to respiratory failure and death.
An in-depth review of the effectiveness of taVNS on the modulation of inflammation was recently published 12. The following information is taken directly from Current Directions in the Auricular Vagus Nerve Stimulation I – A Physiological Perspective (Front Neurosci. 2019;13:854. 2019) 12:
“Inflammation processes are governed through interrelated humoral and neural reflex pathways (Tracey, 2009; Miller and Raison, 2015). In particular, chronic inflammation is based on deregulation of metabolic and immune functions, whereas the imbalance between pro-inflammatory and anti-inflammatory cytokines seems to be decisive in disease progression (Neurath, 2014). Abnormal and chronic inflammation is implicated in, causes and advances, numerous wide-spread chronic diseases as diabetes mellitus and is, for example, a major hindering factor in effective neuroprotection in the brain, e.g., after stroke.
The vagus nerve (VN) provides a first-line defense against infection and inflammation in the periphery to restore homeostasis via conducting information to/from the brain to regulate the immune system. VN is a major component of the neuroendocrine-immune axis (Bonaz and Pellissier, 2016). For instance, even fever, as a brain-mediated response, is signaled to the brain via afferent VN responding to peripheral proinflammatory cytokines, in addition to blood-borne routes for the fever’s signaling (Hansen et al., 2001).
The parasympathetic outflow along VN, i.e., activation of the parasympathetic system, has only anti-inflammatory effects. This was shown by the inverse relationship between VN-mediated parasympathetic markers of the heart rate variability (HRV) and inflammatory markers (Thayer and Fischer, 2008). In contrast, the sympathetic nervous system may have both pro-inflammatory and anti-inflammatory effects.
In general, VN is involved in mainly three reflex pathways with a clear anti-inflammatory role:
- The anti-inflammatory hypothalamic-pituitary-adrenal axis. Here afferent VN fibers sense the level and location of injury/infection in that pro-inflammatory cytokines and/or endotoxins activate VN endings. Somatotopic maps in NTS become activated. Consequently, special neurons in hypothalamus activate the release of hormone adreno-corticotrophin by the hypophysis, stimulating the release of glucocorticoids by the adrenal glands to decrease peripheral inflammation.
- The anti-inflammatory vago-vagal reflex, known also as the cholinergic anti-inflammatory pathway (Borovikova et al., 2000; Tracey, 2007). Here infection-activated afferent VN fibers synapse with and generate an outflow along efferent VN fibers releasing acetylcholine at their synaptic endings. The acetylcholine binds to surface receptors of macrophages and suppresses the production and release of pro-inflammatory cytokines by these macrophages. Interestingly, the tonic neural activity of this cholinergic anti-inflammatory pathway is essential because, when it is impaired, over-inflammation results with an unrestrained cytokine release damaging tissue (Mercante et al., 2018a). From this perspective, vagus nerve stimulation (VNS) enhances the activity of immune-related neural circuits and confers protection of the human body.
- The splenic sympathetic anti-inflammatory pathway. Here the infection-activated afferent VN yields outflow along the efferent VN which stimulates the adrenergic sympathetic nerve in the spleen, releasing norepinephrine at its endings. Then norepinephrine binds to splenic lymphocytes and leads to acetylcholine release by lymphocytes, whereas acetylcholine, in turn, inhibits the release of pro-inflammatory cytokines by splenic macrophages.
Thus, the innate immune system is subjected to a closed-loop reflex modulation via afferent VN fibers, as illustrated in Figure 2A, whereas the activity of the efferent VN maintains homeostasis by limiting pro-inflammatory responses and avoiding immunosuppression. For instance, the role of VN is proposed in informing the brain about peripheral inflammation related to coronary artery disease and in actively modulating the disease related inflammation (Gidron et al., 2006).
An artificial VNS has been shown to harness this natural reflex (Figure 2C). The modulation of VN results in decreased pro-inflammatory and increased anti-inflammatory cytokines, which is effective in suppression of over-inflammation, prevention of tissue injury, and improved survival. For instance, aVNS reduced pro-inflammatory cytokines, as shown in a clinical trial (Stavrakis et al., 2015), and increased norepinephrine levels, as reviewed in Beekwilder and Beems (2010), which supports anti-inflammatory aVNS effects.
In particular, VNS was shown to rebalance the working point of autonomic regulation of the immune system into a protective range avoiding pro-inflammatory responses and, on the other hand, avoiding immunosuppression (Tracey, 2009). Here the working point is defined as the magnitude of innate immune responses relative to the infection or injury stimulus. Chronic changes can unfavorably increase or decrease the working point with the resulting overshooting immune response (with tissue damage, sepsis, or even death) or immunosuppression (with secondary infections), respectively.
In animals, VNS had favorable effects on rheumatoid arthritis in rats (Koopman et al., 2017). VNS reduced surgery-induced intestinal inflammation and improved postoperative intestinal transit in mice, supporting the anti-inflammatory effect of VNS (Matteoli et al., 2013). In addition, VNS prevented the development of shock in rats through inhibited synthesis of the tumor necrosis factor (cytokines) (Borovikova et al., 2000). aVNS was shown to be efficient in mice with lethal endotoxemia or polymicrobial sepsis while reducing systemic tumor necrosis factor due to anti-inflammatory aVNS effects (Huston et al., 2007). aVNS (auricular vagus nerve stimulation) suppressed lipopolysaccharide-induced inflammatory responses in endotoxemic rats through reduced pro-inflammatory cytokines, indicating that aVNS modulates the immune function through the cholinergic anti-inflammatory pathway (Zhao et al., 2012).
In humans, potential therapeutic applications of aVNS are related to chronic inflammatory conditions. These are rheumatoid arthritis, see a clinical trial in Becker (2007), inflammatory bowel disease (Crohn’s disease, ulcerative colitis), and postoperative ileus in order to restore intestinal homeostasis, as reviewed in Tracey (2007), Marshall et al. (2015), and Bonaz and Pellissier (2016).”
Limitations and safety of transcutaneous auricular vagus nerve stimulation
An in-depth review of the limitations of taVNS was recently published. The following is taken directly from Current Directions in the Auricular Vagus Nerve Stimulation I – A Physiological Perspective (Front Neurosci. 2019;13:854. 2019) 12:
“A few indirect but rather seldom unwanted effects can also be triggered by aVNS (auricular vagus nerve stimulation) due to afferent-efferent vagal reflexes, with NTS as a potential intermediate stage. The Arnolds ear-cough reflex is the most dominant reflex, in which mechanical irritation of the auricular skin with embedded aVN (auricular vagus nerve) may cause cough. Other reflexes are ear-gag reflex, ear-lacrimation reflex, ear-syncope reflex, and vaso-vagal reflex. These vegetative reflexes can occur with the respective incidence up to a few percent in the general population (Tekdemir et al., 1998; Ellrich, 2011; Napadow et al., 2012).
In the transcutaneous aVNS, relatively large surface electrodes yield diffuse stimulation fields. Therefore, not only aVN but also other non-vagal fibers in the ear can be expected to be stimulated (Figure 1A). Relatively strong currents and good electrode contacts are required for the current stimuli to circumvent the skin barrier of the ear and still stay suprathreshold in regions innervated by aVN; however, the transcutaneous aVNS is considered as safe (Badran et al., 2018b). The remaining side effects are mostly minor – as related to invasive VNS (Liporace et al., 2001) – and may include headache, pain and skin irritation at the stimulation site, and dizziness (Mertens et al., 2018). In contrast to the transcutaneous aVNS, the auricular needle electrodes in the percutaneous aVNS and the resulting focused stimulation favor precise and local stimulation of aVN endings. Here the electrode contact impedance is lower and more reproducible, favoring a low current stimulation. Minor side effects are local skin irritation, local bleeding, local pain, and dizziness. The clinical incidence of skin irritation and inadvertent bleeding can be reduced down to only 0.05% using a transillumination technique of the ear (Kaniusas et al., 2011), which visualizes auricular vessels to avoid random placement of needles (Roberts et al., 2016).
A special recognition is required on the recent controversy on whether the stimulation effects of aVNS are due to the recruitment of aVN and on the true anatomical location of aVN. The literature on the definite innervation of the auricle is very sparse and is usually based on often cited findings in Peuker and Filler (2002), which unfortunately show some inconsistencies with respect to aVN innervation regions (Burger and Verkuil, 2018). These uncertainties impede a proper interpretation of stimulation effects while an optimal target for aVNS is still under debate (Badran et al., 2018a; Burger and Verkuil, 2018).
Auricular vagus nerve stimulation is typically performed at the tragus or (cavum, cymba) concha (Yakunina et al., 2016). However, some approaches cover larger areas of the auricle (Sator-Katzenschlager and Michalek-Sauberer, 2007) with the potential to stimulate concomitantly a few more auricular nerves in addition to aVN, especially the great auricular nerve (with connections to the spinal cord) or the auriculotemporal nerve (connecting to the nucleus spinalis of the trigeminal nerve). Due to a possible co-activation of the great auricular nerve, it may make sense to study in future the activation of spinal cord sites, e.g., dorsal horn activity (Deuchars et al., 2017). However, current imaging studies in humans are usually focused on VN-activated brain stem nuclei and their projections (Yakunina et al., 2016). In rats, as reported recently in Mahadi et al. (2019), tracing of the transcutaneous stimulation at the tragus labeled the dorsal horn of the cervical spinal cord. Here, a central sympathoinhibition by up to 36% was observed in response to the stimulation, as mediated at least in part through sensory afferent projections to the spinal cord and despite of only sparse labeling of NTS, the termination site of aVN (Figure 1A). Authors in Mahadi et al. (2019) also suggest that the tragus stimulation can indirectly influence brainstem regions involved in the sympathetic control via the spinal cord and even suggest an indirect innervation of NTS by recruited aVN via the spinal cord.
It is questioned if the tragus includes aVN endings or only non-vagal endings, such as the great auricular nerve and the auriculotemporal nerve (Badran et al., 2018a; Burger and Verkuil, 2018). A potential recruitment of these nerves would suggest that mechanisms may be involved for tragal stimulation beyond those anticipated for the sole aVN stimulation. Only the cymba concha was found so far to be solely innervated by aVN (Peuker and Filler, 2002) with the associated maximum activation of vagal projections in NTS during stimulation, as compared to tragus, cavum concha, or earlobe stimulation (Yakunina et al., 2016). However, the cymba concha offers some disadvantages in terms of complexity of electrical stimulation by requiring to insert and/or hold an electrode against the concha, as opposed to having to clip onto the tragus.
Lastly, laterality of aVNS is debated. The stimulation of the left or right aVN cannot be expected to yield different physiological effects since afferent information from both sides are centrally merged in the brainstem (Chen et al., 2015b), and the right and left aVN show comparable counts of Aβ fibers (Safi et al., 2016).
This is in clear contrast to the invasive cervical VNS with dominant lateral effects, in which, for instance, the right-side stimulation recruits predominantly the sinoatrial node (e.g., with the associated bradycardia) and the left side the atrioventricular node. However, simultaneous activation of the left and right aVN may potentially boost stimulation effects due to increased sensory input to the brainstem.”
References
- Wang Y, Dong C, Hu Y, et al. Temporal Changes of CT Findings in 90 Patients with COVID-19 Pneumonia: A Longitudinal Study [published online ahead of print, 2020 Mar 19]. Radiology. 2020;200843.
- Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China [published online ahead of print, 2020 Feb 7]. JAMA. 2020;e201585.
- Chen G et al. Clinical and immunologic features in severe and moderate Coronavirus Disease 2019. J Clin Invest. 2020 Mar 27. Pii: 137244.
- Zheng HY et al. Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cell Mol Immunol. 2020 Mar 17.
- Sun F, Kou H, Wang S, et al. Medication patterns and disease progression among 165 patients with coronavirus disease 2019 (COVID-19) in Wuhan, China: a single-centered, retrospective, observational study. Preprints with the Lancet. 2020. [Preprint].
- Mehta et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020 Mar 28;395(10229):1033-1034. Mar 16.
- Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395(10223):473-475.
- Ritchie AI, Singanayagam A. Immunosuppression for hyperinflammation in COVID-19: a double-edged sword?. Lancet. 2020;395(10230):1111.
- Bussalino E, De Maria A, Russo R, Paoletti E. Immunosuppressive therapy maintenance in a kidney transplant recipient SARS-CoV-2 pneumonia: a case report [published online ahead of print, 2020 Apr 13]. Am J Transplant. 2020;10.1111/ajt.15920.
- Spezzani V, Piunno A, Iselin HU. Benign COVID-19 in an immunocompromised cancer patient – the case of a married couple. Swiss Med Wkly. 2020;150:w20246. Published 2020 Apr 11.
- Tanaka S, Hammond B, Rosin DL, Okusa MD. Neuroimmunomodulation of tissue injury and disease: an expanding view of the inflammatory reflex pathway. Bioelectron Med. 2019;5:13. Published 2019 Aug 13.
- Kaniusas E, Kampusch S, Tittgemeyer M, et al. Current Directions in the Auricular Vagus Nerve Stimulation I – A Physiological Perspective. Front Neurosci. 2019;13:854. Published 2019 Aug 9.
- Koopman FA et al. Vagus nerve stimulation inhibits cytokine production and attenuates disease severity in rheumatoid arthritis. Proc Natl Acad Sci U S A. 2016;113(29):8284–8289.
- de Jonge WJ, van der Zanden EP, The FO, et al. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway [published correction appears in Nat Immunol. 2005 Sep;6(9):954]. Nat Immunol. 2005;6(8):844–851.
- Zhao YX et al. Transcutaneous auricular vagus nerve stimulation protects endotoxemic rat from lipopolysaccharide-induced inflammation. Evid Based Complement Alternat Med. 2012;2012:627023.
- Stavrakis S, Humphrey MB, Scherlag B, et al. Low-Level Vagus Nerve Stimulation Suppresses Post-Operative Atrial Fibrillation and Inflammation: A Randomized Study. JACC Clin Electrophysiol. 2017;3(9):929–938.
- Yamakawa K, Matsumoto N, Imamura Y, et al. Electrical vagus nerve stimulation attenuates systemic inflammation and improves survival in a rat heatstroke model. PloS One. 2013;8(2):e56728.
- Huston JM et al. Transcutaneous vagus nerve stimulation reduces serum high mobility group box 1 levels and improves survival in murine sepsis. Crit Care Med. 2007 Dec;35(12):2762-8.
- Bonaz B, Sinniger V, Hoffmann D, et al. Chronic vagus nerve stimulation in Crohn’s disease: a 6-month follow-up pilot study. Neurogastroenterol Motil. 2016;28(6):948–953.
- Liu A, Rong P, Gong L, et al. Efficacy and Safety of Treatment with Transcutaneous Vagus Nerve Stimulation in 17 Patients with Refractory Epilepsy Evaluated by Electroencephalogram, Seizure Frequency, and Quality of Life. Med Sci Monit. 2018;24:8439–8448. Published 2018 Nov 23.
- 21. NIH Coronavirus Disease 2019 (COVID-19) Treatment Guidelines, accessed April 23, 2020 at https://covid19treatmentguidelines.nih.gov/.
- Redgrave J, Day D, Leung H, et al. Safety and tolerability of Transcutaneous Vagus Nerve stimulation in humans; a systematic review. Brain Stimul. 2018;11(6):1225–1238.
- dos Santos CC, Shan Y, Akram A, Slutsky AS, Haitsma JJ. Neuroimmune regulation of ventilator-induced lung injury. Am J Respir Crit Care Med. 2011;183(4):471–482.
- Wu H, Li L, Su X. Vagus nerve through α7 nAChR modulates lung infection and inflammation: models, cells, and signals. Biomed Res Int. 2014;2014:283525.
- van Westerloo DJ et al. The cholinergic anti-inflammatory pathway regulates the host response during septic peritonitis. J Infect Dis.2005 Jun 15;191(12):2138-48. Epub 2005 May 10.
- Noé E, Ferri J, Colomer C, et al. Feasibility, safety and efficacy of transauricular vagus nerve stimulation in a cohort of patients with disorders of consciousness. Brain Stimul. 2020;13(2):427–429.
- Kreuzer PM, Landgrebe M, Husser O, et al. Transcutaneous vagus nerve stimulation: retrospective assessment of cardiac safety in a pilot study. Front Psychiatry. 2012;3:70. Published 2012 Aug 7.
- Redgrave JN, Moore L, Oyekunle T, et al. Transcutaneous Auricular Vagus Nerve Stimulation with Concurrent Upper Limb Repetitive Task Practice for Poststroke Motor Recovery: A Pilot Study. J Stroke Cerebrovasc Dis. 2018;27(7):1998–2005.
- Kilgard MP, Rennaker RL, Alexander J, Dawson J. Vagus nerve stimulation paired with tactile training improved sensory function in a chronic stroke patient. NeuroRehabilitation. 2018;42(2):159–165.
- Conlon B, Hamilton C, Hughes S, et al. Noninvasive Bimodal Neuromodulation for the Treatment of Tinnitus: Protocol for a Second Large-Scale Double-Blind Randomized Clinical Trial to Optimize Stimulation Parameters. JMIR Res Protoc. 2019;8(9):e13176. Published 2019 Sep 27.
- Bretherton B, Atkinson L, Murray A, Clancy J, Deuchars S, Deuchars J. Effects of transcutaneous vagus nerve stimulation in individuals aged 55 years or above: potential benefits of daily stimulation. Aging (Albany NY). 2019;11(14):4836–4857.
32. Borges U, Laborde S, Raab M. Influence of transcutaneous vagus nerve stimulation on cardiac vagal activity: Not different from sham stimulation and no effect of stimulation intensity. PloS One. 2019;14(10):e0223848. Published 2019 Oct 11.